ATB-352, a potent and non-addictive analgesic, targets the $9 billion market for post-operative pain.
ATB-352 is a hydrogen sulfide-releasing derivative of ketoprofen, one of the most potent NSAIDs prescribed for acute pain. However, ketoprofen is also one of the most damaging NSAIDs to the gastrointestinal tract. ATB-352 has been shown to be gastrointestinal-safe and non-addictive in animal studies. Investigational New Drug-enabling studies for ATB-352 are underway, with Phase 1 human trials slated to begin in late 2020.
Global Opioid Crisis
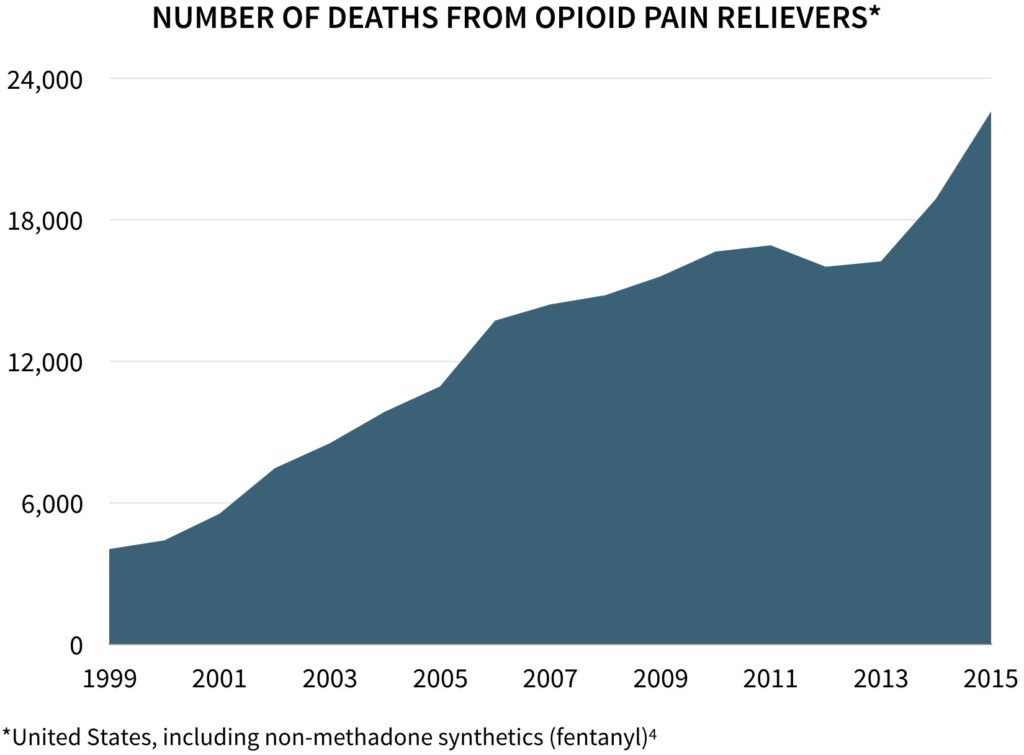
While opioids (e.g., oxycodone, fentanyl) are widely prescribed for acute pain relief, they are often abused due to their highly addictive nature. More than 60% of drug overdose deaths involve an opioid. With total mortality having quadrupled since 1999, more deaths are now attributable to opioids than to road accidents.
“On average, 130 Americans die every day from an opioid overdose.”
– Centers for Disease Control and Prevention
The misuse of prescription opioids costs the United States nearly $80 billion per year, including the costs of healthcare, lost productivity, addiction treatment, and criminal justice involvement.1
There is a widely recognized, urgent need for a safer, non-addictive pain-killer for post-operative pain.
Lead Indication: Post-operative Pain
The over-prescription of opioids is being driven largely by the lack of alternative therapies that are both safe and effective. Of the 50 million US surgical patients requiring post-operative pain medication each year, 80% are prescribed opioids, with more than 6% becoming persistent opioid users after surgery. ATB-352 represents a compelling therapy for surgery patients given its analgesic potency, gastrointestinal safety and lack of abuse potential.
Many surgeries require a multi-day hospital recovery period, during which intravenous agents are often prescribed to manage post-operative pain. Upon discharge from hospital care, patients may continue to suffer from pain for several weeks. This transition can be challenging for both patients and physicians, who seek to prescribe an oral therapeutic that is effective but does not create addiction concerns. To address this, Antibe has adopted a two-stage product strategy to streamline management of post-operative pain: (i) an aqueous formulation for intravenous administration of ATB-352 while in hospital; and (ii) a tablet formulation for use following hospital discharge.
In view of ATB-352’s potential to replace opioid use, Antibe will pursue an FDA Fast Track designation to expedite the development and regulatory approval process.
ATB-352: Non-addictive and Gastrointestinal-safe
In preclinical studies, ATB-352 caused negligible intestinal damage compared to ketoprofen, despite comparable suppression of intestinal prostaglandin or whole blood thromboxane synthesis.2 The rats were treated orally twice-daily for 5 days with either 10 mg/kg ketoprofen, or an equimolar dose of ATB-352 (14.5 mg/kg). Pharmacokinetic and toxicology studies suggest that ATB-352, like ATB-346, is effective when administered once daily.3
When tested over a concentration range of 3 to 30 µg/mL in a CEREP in vitro assay, no agonist effect of ATB-352 was found against the µ(MOP) opioid receptor, indicating no addiction potential.3
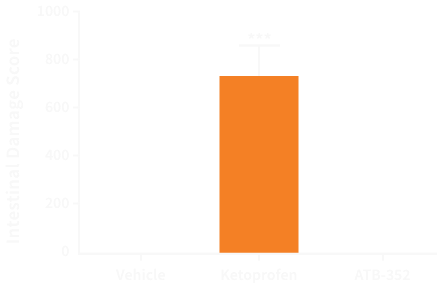
ATB-352 causes negligible gastrointestinal damage in rats compared to ketoprofen, a very strong NSAID prescribed for acute pain.
(1) Florence et al. The Economic Burden of Prescription Opioid Overdose, Abuse, and Dependence in the United States, 2013.
Med Care. 2016;54(10):901-906.
(2) Gemici et al. H2S-releasing drugs: anti-inflammatory, cytoprotective and chemopreventative potential. Nitric Oxide. 2015 Apr 30;46:25-31.
(3) Antibe pre-clinical data on ATB-352.
